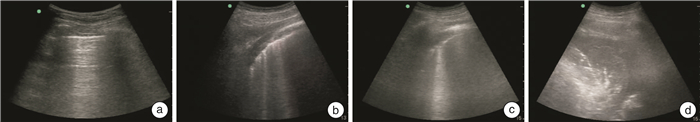
Clinical value of lung ultrasound combined with neutrophil to lymphocyte ratio and D-Dimer in evaluating the efficacy and prognosis of hospital-acquired pneumonia
-
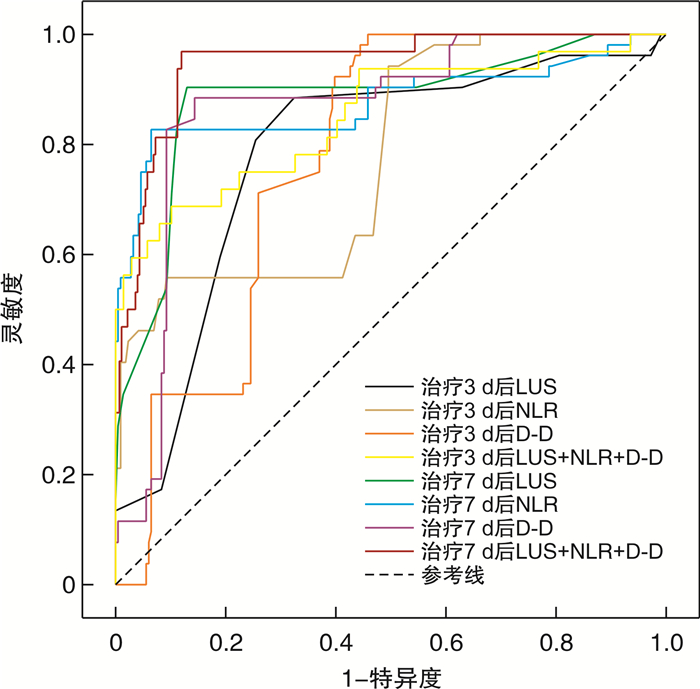
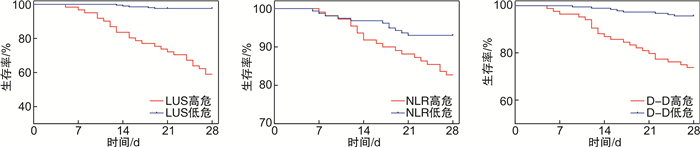
摘要: 目的 探讨肺部超声、中性粒细胞与淋巴细胞比值(NLR)、D-二聚体(D-D)及三者联合在医院获得性肺炎(HAP)的治疗效果和预后评价方面的应用价值。方法 回顾性选取2019年1月—2022年3月在北京华信医院重症医学科和急诊科接受治疗的268例HAP患者,按照疗效的区别进行分组,分为有效组与无效组,有效组216例,无效组52例。分别对2组患者的一般资料以及治疗开始前、治疗3 d后、治疗7 d后的D-D、外周血NLR与肺部超声评分(LUS)的变化进行比较; 通过Pearson相关性分析对上述指标和临床肺部感染评分(CPIS)之间的关联性进行分析; 通过logistic回归方程对影响疗效的有关因素进行分析; 通过受试者工作特征(ROC)曲线与ROC曲线下面积(AUC)对以上3个指标及三者联合的疗效预测效能进行分析; 通过Pearson相关性分析对上述指标和序贯器官衰竭估计评分(SOFA)之间的关联性进行分析; 采用Kaplan-Meier曲线分析LUS、NLR、D-D高危组与低危组患者的生存率。结果 有效组治疗3 d及7 d后的LUS、NLR、D-D均低于无效组(P < 0.05);经Pearson线性相关性分析发现治疗7 d后的LUS、NLR及D-D均与CPIS呈正相关(P < 0.05),其中治疗7 d后的LUS与CPIS的相关性最强(r=0.532);logistic回归方程计算结果显示,治疗3 d与治疗7 d后的D-D、外周血NLR以及LUS都和HAP具备关联性(P < 0.05);ROC曲线分析,治疗7 d后LUS、NLR、D-D预测HAP治疗无效的AUC大于治疗3 d的AUC,治疗7 d后的LUS、NLR、D-D三者联合预测疗效的AUC最大,AUC=0.913;Pearson相关性分析显示治疗7 d后的LUS、NLR及D-D均与SOFA评分呈正相关(P < 0.05),其中治疗7 d后的LUS与SOFA评分的相关性最强(r=0.525)。Kaplan-Meier曲线分析发现,治疗7 d后LUS、NLR、D-D高危与低危患者生存率比较,差异均有统计学意义(P < 0.05)。结论 肺部超声联合NLR及D-D指标监测对评价HAP的疗效和预后具有临床价值,值得推广研究。Abstract: Objective To discuss the application value of lung ultrasound, neutrophil to lymphocyte ratio(NLR), D-dimer(D-D) and their combination in the treatment and prognosis evaluation of hospital acquired pneumonia(HAP).Methods A total of 268 patients with HAP who were treated in the intensive care unit and emergency department of the Beijing Huaxin Hospital from January 2019 to March 2022 were reviewed and analyzed. The patients were divided into effective group and ineffective group, with 216 in effective group and 52 in ineffective group, according to the difference of curative effect. The general data of the two groups were compared, and the changes of D-D, peripheral blood NLR and lung ultrasonic score(LUS) before treatment, 3 days after treatment and 7 days after treatment were compared. Pearson correlation was used to analyze the correlation between the above indicators and clinical pulmonary infection score(CPIS); Logistic regression equation was used to analyze the factors affecting the curative effect. The above three indicators and their combined efficacy prediction were analyzed through the receiver operating characteristic(ROC) and area under ROC curve(AUC) of patients participating in the experiment. Pearson correlation was used to analyze the correlation between the above indicators and sequential organ failure assessment(SOFA); Kaplan-Meier survival curve was used to analyze the survival rate of patients with high-risk and low-risk LUS, NLR and D-D.Results In the effective group, the LUS, NLR and D-D after 3 days and 7 days of treatment were lower than those of the ineffective group. Pearson linear correlation analysis showed that LUS, NLR and D-D after 7 days of treatment were positively correlated with CPIS score(P < 0.05), among which LUS after 7 days of treatment had the strongest correlation with CPIS(r=0.532). Logistic regression analysis showed that LUS, NLR and D-D were correlated with the efficacy of HAP after 3 days and 7 days of treatment(P < 0.05). ROC curve analysis showed that the AUC of LUS, NLR and D-D predicting invalid HAP after 7 days of treatment was higher than that after 3 days of treatment. The AUC of LUS combined with NLR and D-D after 7 days of treatment to predict the efficacy was the largest, AUC=0.913. Pearson correlation analysis showed that LUS, NLR and D-D after 7 days of treatment were positively correlated with SOFA scores(P < 0.05), and LUS after 7 days of treatment had the strongest correlation with SOFA scores(r=0.525). Kaplan-Meier curve analysis showed that after 7 days of treatment, the survival rates of LUS, NLR, D-D high-risk and low-risk patients were significantly different(P < 0.05).Conclusion Pulmonary ultrasound combined with NLR and D-D dynamic monitoring has clinical application value in evaluating the efficacy and prognosis of HAP, and is worthy of promotion and research.
-
Key words:
- ultrasound /
- lung /
- neutrophil to lymphocyte ratio /
- D-Dimer /
- hospital acquired pneumonia /
- curative effect /
- prognosis
-

-
表 1 2组患者基线资料比较
X±S,例(%) 资料 有效组
(n=216)无效组
(n=52)t/χ2 P 年龄/岁 0.041 0.840 ≤60 78(36.11) 18(34.62) >60 138(63.89) 34(65.38) 性别 0.101 0.750 男 111(51.39) 28(53.85) 女 105(48.61) 24(46.15) 吸烟史 72(33.33) 15(28.85) 0.385 0.535 基础疾病 脑梗死 31(12.96) 12(23.08) 3.376 0.066 糖尿病 43(19.91) 12(23.08) 0.258 0.611 心功能不全 36(16.67) 15(28.85) 3.750 0.053 原发性高血压 58(26.85) 18(34.62) 0.035 0.852 HAP发生科室 1.072 0.585 养老及康复机构 108(50.00) 27(51.92) 本院内科及外科 66(30.56) 18(34.62) 本院急诊及ICU 42(19.44) 7(13.46) 插管机械通气 0.129 0.720 是 57(26.39) 13(25.00) 否 159(73.61) 39(75.00) HAP病原体分类 0.528 0.768 细菌 168(77.78) 38(73.08) 真菌 28(12.96) 8(15.38) 其他 20(9.26) 6(11.54) 氧合指数 223.5± 0.884 226.1± 1.433 1.303 0.193 CPIS 8.537± 0.090 8.519± 0.179 0.088 0.930 APACHE Ⅱ评分/分 17.480± 0.183 18.000± 0.709 1.078 0.282 表 2 2组患者LUS、NLR、D-D动态变化
X±S 组别 例数 LUS/分 NLR D-D/(mg·L-1) 治疗前 治疗3 d后 治疗7 d后 治疗前 治疗3 d后 治疗7 d后 治疗前 治疗3 d后 治疗7 d后 有效组 216 13.130± 0.144 12.150± 0.143 9.361± 0.188 4.592± 0.085 3.033± 0.076 2.358± 0.046 1.299± 0.059 1.176± 0.054 0.916± 0.047 无效组 52 13.330± 0.309 14.460± 0.286 15.370± 0.416 4.994± 0.377 5.269± 0.339 6.147± 0.205 1.435± 0.030 1.897± 0.035 2.134± 0.036 t 0.599 7.148 13.830 1.589 9.860 27.630 1.120 6.418 11.970 P 0.550 <0.001 <0.001 0.113 <0.001 <0.001 0.264 <0.001 <0.001 表 3 疗效影响因素的logistic回归分析
影响因素 SE β OR 95%CI P 治疗3 d后 LUS 0.166 0.208 1.231 0.889~1.721 < 0.001 NLR 0.343 0.334 1.397 0.821~3.409 < 0.001 D-D 1.084 0.437 1.548 0.169~12.34 < 0.001 治疗7 d后 LUS 0.101 0.392 1.479 1.234~1.843 < 0.001 NLR 0.165 0.724 2.062 1.528~2.953 < 0.001 D-D 0.623 1.495 4.457 1.451~16.99 < 0.001 -
[1] 中华医学会呼吸病学分会感染学组. 中国成人医院获得性肺炎与呼吸机相关性肺炎诊断和治疗指南(2018年版)[J]. 中华结核和呼吸杂志, 2018, 41(4): 255-280. doi: 10.3760/cma.j.issn.1001-0939.2018.04.006
[2] Renaud C, Kollef MH. Classical and molecular techniques to diagnose HAP/VAP[J]. Semin Respir Crit Care Med, 2022, 43(2): 219-228. doi: 10.1055/s-0041-1739359
[3] 胡才宝, Lichtenstein DA. 重症肺部超声的过去、现在与未来[J]. 中华诊断学电子杂志, 2018, 6(2): 77-79. https://www.cnki.com.cn/Article/CJFDTOTAL-ZDDZ201802002.htm
[4] Zahorec R. Neutrophil-to-lymphocyte ratio, past, present and future perspectives[J]. Bratisl Lek Listy, 2021, 122(7): 474-488.
[5] Soni M, Gopalakrishnan R, Vaishya R, et al. D-dimer level is a useful predictor for mortality in patients with COVID-19: analysis of 483 cases[J]. Diabetes Metab Syndr, 2020, 14(6): 2245-2249. doi: 10.1016/j.dsx.2020.11.007
[6] Manchal N, Siriwardena M, Hay K, et al. Assessment of the Clinical Pulmonary Infection Scores for prediction of ventilator associated pneumonia in patients with out of hospital cardiac arrest[J]. Infect Dis Health, 2021, 26(1): 48-54. doi: 10.1016/j.idh.2020.09.001
[7] Zou XJ, Li SS, Fang MH, et al. Acute physiology and chronic health evaluation Ⅱ score as a predictor of hospital mortality in patients of coronavirus disease 2019[J]. Crit Care Med, 2020, 48(8): e657-e665. doi: 10.1097/CCM.0000000000004411
[8] Srinivasan S, Aggarwal N, Makhaik S, et al. Role of lung ultrasound in diagnosing and differentiating transient tachypnea of the newborn and respiratory distress syndrome in preterm neonates[J]. J Ultrason, 2022, 22(88): e1-e5. doi: 10.15557/JoU.2022.0001
[9] Allinovi M, Parise A, Giacalone M, et al. Lung ultrasound may support diagnosis and monitoring of COVID-19 pneumonia[J]. Ultrasound Med Biol, 2020, 46(11): 2908-2917. doi: 10.1016/j.ultrasmedbio.2020.07.018
[10] Modi AR, Kovacs CS. Hospital-acquired and ventilator-associated pneumonia: diagnosis, management, and prevention[J]. Cleve Clin J Med, 2020, 87(10): 633-639. doi: 10.3949/ccjm.87a.19117
[11] Pölkki A, Pekkarinen PT, Takala J, et al. Association of Sequential Organ Failure Assessment(SOFA)components with mortality[J]. Acta Anaesthesiol Scand, 2022, 66(6): 731-741. doi: 10.1111/aas.14067
[12] Wicky PH, Martin-Loeches I, Timsit JF. HAP and VAP after guidelines[J]. Semin Respir Crit Care Med, 2022, 43(2): 248-254. doi: 10.1055/s-0041-1740246
[13] Liu JY, Dickter JK. Nosocomial infections: a history of hospital-acquired infections[J]. Gastrointest Endosc Clin N Am, 2020, 30(4): 637-652. doi: 10.1016/j.giec.2020.06.001
[14] 王书鹏, 段军. 重症肺部超声临床应用管理流程[J]. 中华诊断学电子杂志, 2018, 6(2): 98-100. https://www.cnki.com.cn/Article/CJFDTOTAL-ZDDZ201802008.htm
[15] Staub LJ, Mazzali Biscaro RR, Kaszubowski E, et al. Lung ultrasound for the emergency diagnosis of pneumonia, acute heart failure, and exacerbations of chronic obstructive pulmonary disease/asthma in adults: a systematic review and meta-analysis[J]. J Emerg Med, 2019, 56(1): 53-69. doi: 10.1016/j.jemermed.2018.09.009
[16] Bar A, Lin G, Lazar LO, et al. Immediate pneumothorax diagnosis by surgical residents using portable ultrasound[J]. Innovations(Phila), 2021, 16(2): 152-156. doi: 10.1177/1556984520978315
[17] Hassanzad M, Kiani A, Abedini A, et al. Lung ultrasound for the diagnosis of cystic fibrosis pulmonary exacerbation[J]. BMC Pulm Med, 2021, 21(1): 353. doi: 10.1186/s12890-021-01728-8
[18] Hansell L, Milross M, Delaney A, et al. Lung ultrasound has greater accuracy than conventional respiratory assessment tools for the diagnosis of pleural effusion, lung consolidation and collapse: a systematic review[J]. J Physiother, 2021, 67(1): 41-48. doi: 10.1016/j.jphys.2020.12.002
[19] Unlukaplan IM, Dogan H, Ozucelik DN. Lung ultrasound for the diagnosis of pneumonia in adults[J]. J Pak Med Assoc, 2020, 70(6): 989-992.
[20] Trias-Sabrià P, Molina-Molina M, Aso S, et al. Lung ultrasound score to predict outcomes in COVID-19[J]. Respir Care, 2021, 66(8): 1263-1270. doi: 10.4187/respcare.08648
[21] 袁雪梅, 韩小琴, 韦梅, 等. 早期肝素结合蛋白联合降钙素原对重症肺炎患者的预后评估价值[J]. 临床急诊杂志, 2022, 23(8): 553-556. https://lcjz.whuhzzs.com/article/doi/10.13201/j.issn.1009-5918.2022.08.003
[22] Huang ZW, Fu ZY, Huang WJ, et al. Prognostic value of neutrophil-to-lymphocyte ratio in sepsis: a meta-analysis[J]. Am J Emerg Med, 2020, 38(3): 641-647. doi: 10.1016/j.ajem.2019.10.023
[23] Zhou YQ, Feng DY, Li WJ, et al. Lower neutrophil-to-lymphocyte ratio predicts high risk of multidrug-resistant Pseudomonas aeruginosa infection in patients with hospital-acquired pneumonia[J]. Ther Clin Risk Manag, 2018, 14: 1863-1869.
[24] Zhang LT, Yan XS, Fan QK, et al. D-dimer levels on admission to predict in-hospital mortality in patients with Covid-19[J]. J Thromb Haemost, 2020, 18(6): 1324-1329.
[25] 杨少芬, 李素. 降钙素原、C反应蛋白及D-二聚体在重症肺炎患者中的表达及其与预后的关系[J]. 广东医科大学学报, 2021, 39(3): 284-286. https://www.cnki.com.cn/Article/CJFDTOTAL-GDYY202103010.htm
-




 下载:
下载: