Prognostic value of ultrasonographic measurement of optic nerve sheath diameter combined with Pcv-aCO2/Ca-cvO2 for prehospital mannitol administration in patients with severe traumatic brain injury
-
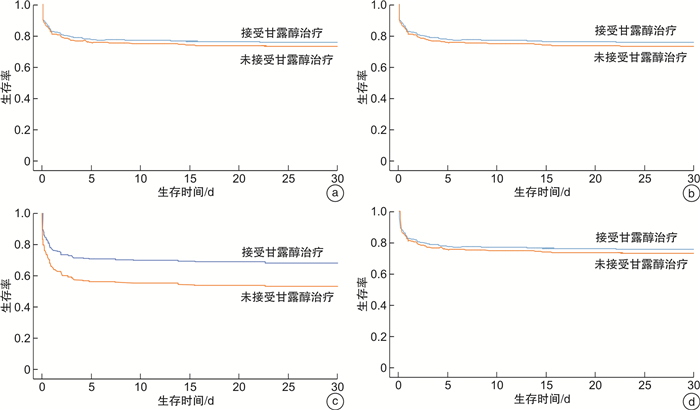
摘要: 目的 床旁超声测量视神经鞘直径(ONSD)判断颅内压(ICP)联合Pcv-aCO2/Ca-cvO2指导重型颅脑外伤(TBI)患者院前甘露醇应用的评估价值。方法 采用前瞻性队列研究选择我院2021年1月-2022年1月院前接诊的重型TBI患者,将其中格拉斯哥昏迷评分(GCS) < 8分者作为研究对象。院前接诊后,通过床旁超声测量ONSD评估ICP,置入中心静脉导管,采取颈内静脉血样本,计算Pcv-aCO2/Ca-cvO2值。将患者分为4组:A组,Pcv-aCO2/Ca-cvO2>1.8且ONSD>5 mm; B组,Pcv-aCO2/Ca-cvO2>1.8且ONSD≤5 mm; C组,Pcv-aCO2/Ca-cvO2≤1.8且ONSD>5 mm; D组,Pcv-aCO2/Ca-cvO2≤1.8且ONSD≤5 mm。研究期间,院前共接诊343例TBI患者,根据纳入、排除标准,最终275例患者纳入分析,其中A组77例,B组69例,C组70例,D组59例。以死亡或出院作为观察终点。观察、记录并分析是否使用甘露醇在4组间30 d病死率、神经功能恢复情况[通过格拉斯哥预后评分(GOS)进行评估]、住院天数差异。结果 在未经调整的logistic回归分析中,C组患者接受甘露醇治疗是重型TBI患者30 d病死率增高的保护性因素(OR=0.36,95%CI:0.15~0.86,P< 0.001)。在调整混杂因素后,C组患者接受甘露醇治疗仍是重型TBI患者30 d病死率增高的保护性因素(OR=0.31,95%CI:0.11~0.73,P=0.004)。其他3组院前使用甘露醇与30 d病死率之间无相关性。Cox生存曲线显示C组患者使用甘露醇后生存率明显提高。结论 ICP监测联合Pcv-aCO2/Ca-cvO2有助于指导院前重型TBI患者甘露醇的使用。
-
关键词:
- 甘露醇 /
- 院前 /
- 重型颅脑创伤 /
- 视神经鞘直径 /
- Pcv-aCO2/Ca-cvO2
Abstract: Objective We aimed to investigate the significance of bedside ultrasonography in measurement of optic nerve sheath diameter(ONSD) combined with central venous-arterial carbon dioxide tension to arterial-central venous oxygen content ratio(Pcv-aCO2/Ca-cvO2) as indication of mannitol administration in patients with severe traumatic brain injury(TBI).Methods A prospective cohort study was conducted. The patients admitted to prehospital of patients with severe traumatic brain injury admitted prehospital from January 2021 to January 2022 and Glasgow coma scale < 8 were selected as the research objects. We try to use ultrasonographic to measure optic nerve sheath diameter for intracranial pressure monitoring and insert the central venous catheter taken the venous blood samples, then calculate the Pcv-aCO2/ Ca-cvO2values. Patients were classified into 4 groups according to Pcv-aCO2/Ca-cvO2 and ONSD: Group A, Pcv-aCO2/Ca-cvO2>1.8, ONSD>5 mm; Group B: Pcv-aCO2/Ca-cvO2>1.8, ONSD≤5 mm; Group C: Pcv-aCO2/Ca-cvO2≤1.8, ONSD>5 mm and Group D: Pcv-aCO2/Ca-cvO2≤1.8, ONSD≤5 mm. Death discharge were regarded as the end of observation. The 30-day mortality, functional neurological recovery at discharge(measured by Glasgow Outcome Scale), and length of hospital stay were compared among groups.Results During the study, 343 patients with severe traumatic brain injury were admitted prehospital. According to the inclusion and exclusion criteria, 275 patients were included in the analysis, including 77 patients in group A, 69 patients in group B, 70 patients in group C and 59 patients in group D. In the unadjusted analysis, lower 30-day mortality was substantially decreased in patients who received prehospital mannitol in group C(OR=0.36, 95%CI: 0.15-0.86,P< 0.001), compared with patients who did not receive prehospital mannitol. After adjustment for confounders, a substantial decrease in the odds of 30-day mortality in patients with severe TBI who received prehospital mannitol(OR=0.31, 95%CI: 0.11-0.73,P=0.004). No association between prehospital administration of mannitol and mortality was found across others group. Survival analyses also consistently showed an increased survival probability in patients with severe TBI in group C after mannitol administration.Conclusion Combination of ICP and Pcv-aCO2/Ca-cvO2ratio are useful for mannitol administration in patients with severe traumatic brain injury. -

-
表 1 接受甘露醇治疗与未接受者基本资料分析
例(%),M(P25,P75) 基本资料和创伤数据 接受甘露醇(131例) 未接受甘露醇(144例) P 年龄/岁 49(28,59) 47(23,67) 0.07 性别 0.08 男 88(67) 94(65) 女 43(33) 50(35) 创伤分类 单纯TBI 67(51) 71(49) 0.06 多发伤 64(49) 73(51) 0.06 ISS评分/分 37(21,58) 26(17,39) < 0.01 院前重要体查参数 GCS评分/分 4(3,6) 8(3,10) < 0.01 SPO2/% 87(81,95) 90(83,97) 0.40 收缩压/mmHg 182(91,201) 149(99,183) 0.03 30 d病死率/% 53(69/131) 40(57/144) < 0.01 出院时GOS评估情况 0.06 死亡 3(2) 5(3) 植物状态 9(7) 14(10) 严重残疾 56(43) 59(41) 中等残疾 23(18) 31(22) 恢复良好 40(31) 35(24) 住院天数/d 16(6,32) 11(7.5,27.0) 0.01 表 2 调整前后使用甘露醇与病死率在4组间的回归分析
组别 未调整的回归分析 调整后的回归分析 OR(95%CI) P OR(95%CI) P A组 1.03(0.97~1.22) 0.06 1.01(0.88~1.19) 0.07 B组 1.02(0.94~1.17) 0.09 0.99(0.95~1.04) 0.09 C组 0.36(0.15~0.86) < 0.001 0.31(0.11~0.73) 0.004 D组 1.09(0.94~1.20) 0.08 1.03(0.91~1.13) 0.07 注:使用logistic回归分析比较4组间重型TBI患者与30 d病死率间的关系; 调整后的多因素logistic回归分析模型,相应控制变量被加入该模型中,包括患者基本资料、创伤类型、创伤前基础情况、创伤严重程度评估、首次生命体征(收缩压、心率、血氧饱和度)等。 -
[1] 吉其舰, 彭海丽, 郭小敏. 重度颅脑外伤患者术后监测颅内压及压力相关指数的临床价值[J]. 临床急诊杂志, 2021, 22(5): 335-339. https://www.cnki.com.cn/Article/CJFDTOTAL-ZZLC202105009.htm
[2] Capizzi A, Woo J, Verduzco-Gutierrez M. Traumatic Brain Injury: An Overview of Epidemiology, Pathophysiology, and Medical Management[J]. Med Clin North Am, 2020, 104(2): 213-238. doi: 10.1016/j.mcna.2019.11.001
[3] Zeiler FA, Aries M, Czosnyka M, et al. Cerebral Autoregulation Monitoring in Traumatic Brain Injury: An Overview of Recent Advances in Personalized Medicine[J]. J Neurotrauma, 2022.
[4] Robinson CP. Moderate and Severe Traumatic Brain Injury[J]. Continuum(Minneap Minn), 2021, 27(5): 1278-1300.
[5] Marklund N, Bellander BM, Godbolt AK, et al. Treatments and rehabilitation in the acute and chronic state of traumatic brain injury[J]. J Intern Med, 2019, 285(6): 608-623. doi: 10.1111/joim.12900
[6] Krishnamoorthy V, Komisarow JM, Laskowitz DT, et al. Multiorgan Dysfunction After Severe Traumatic Brain Injury: Epidemiology, Mechanisms, and Clinical Management[J]. Chest, 2021, 160(3): 956-964. doi: 10.1016/j.chest.2021.01.016
[7] Kochanek PM, Tasker RC, Carney N, et al. Guidelines for the Management of Pediatric Severe Traumatic Brain Injury, Third Edition: Update of the Brain Trauma Foundation Guidelines, Executive Summary[J]. Neurosurgery, 2019, 84(6): 1169-1178. doi: 10.1093/neuros/nyz051
[8] Chesnut RM, Temkin N, Carney N, et al. A trial of intracranialpressure monitoring in traumatic brain injury[J]. N Eng J Med, 2012, 367: 2471-2481. doi: 10.1056/NEJMoa1207363
[9] 日本神经外科学会. 重型头部外伤治疗·管理指南[M]. 第2版. 神经外伤, 2006, 29: 1-115.
[10] GAO XH, Cao W, Li PJ. Pcv-aCO2/Ca-cvO2 Combined With Arterial Lactate Clearance Rate as Early Resuscitation Goals in Septic Shock[J]. Am J Med Sci, 2019, 358(3): 182-190. doi: 10.1016/j.amjms.2019.04.025
[11] Dubin A, Pozo MO, Hurtado J. Central venous minus arterial carbon dioxide pressure to arterial minus central venous oxygen content ratio as an indicator of tissue oxygenation: a narrative review[J]. Rev Bras Ter Intensiva, 2020, 32(1): 115-122.
[12] Berthet M, Durand M. Meaning of the venoarterial carbon dioxide difference/arterial-venous oxygen difference ratio[J]. Crit Care Med, 2013, 41(12): e489-90.
[13] 包赘, 鲍南, 陈文劲, 等. 甘露醇治疗颅内压增高专家共识[J]. 中华医学杂志, 2019, 99(23): 1763-1766. doi: 10.3760/cma.j.issn.0376-2491.2019.23.002
[14] 赵立娜, 谢晖, 王瑞兰. 超声测量视神经鞘直径评估颅内压增高的临床研究进展[J]. 中华重症医学电子杂志(网络版), 2020, 6(1): 113-116. https://www.cnki.com.cn/Article/CJFDTOTAL-ZZYD202001033.htm
[15] Stevens R, Gommer ED, Aries M, et al. Optic nerve sheath diameter assessment by neurosonology: A review of methodologic discrepancies[J]. J Neuroimaging, 2021, 31(5): 814-825. doi: 10.1111/jon.12906
[16] Godoy DA, Rabinstein AA. How to manage traumatic brain injury without invasive monitoring?[J]. Curr Opin Crit Care, 2022, 28(2): 111-122. doi: 10.1097/MCC.0000000000000914
[17] 王蕾, 朱保锋, 张毅, 等. 急诊头痛患者视神经鞘直径与颅内压相关性研究[J]. 临床急诊杂志, 2020, 21(1): 25-28, 33. https://www.cnki.com.cn/Article/CJFDTOTAL-ZZLC202001002.htm
[18] Mukai A, Suehiro K, Kimura A, et al. Comparison of the venous-arterial CO2 to arterial-venous O2 content difference ratio with the venous-arterial CO2 gradient for the predictability of adverse outcomes after cardiac surgery[J]. Clin Monit Comput, 2020, 34(1): 41-53. doi: 10.1007/s10877-019-00286-z
[19] Zante B, Kluge S. Central Venous-Arterial CO2 Difference in Cardiac Surgery Patients-A Parameter in Relationship to Cardiac Output and Altered Microcirculatory Blood Flow[J]. Shock, 2020, 54(2): 273-274. doi: 10.1097/SHK.0000000000001525
[20] Molloy S, Batchelor G, Mallett P, et al. Fifteen-minute consultation: Severe traumatic brain injury in paediatrics[J]. Arch Dis Child Educ Pract Ed, 2021, 106(1): 9-17. doi: 10.1136/archdischild-2019-318246
[21] Petkus V, Preiksaitis A, Chaleckas E, et al. Optimal Cerebral Perfusion Pressure: Targeted Treatment for Severe Traumatic Brain Injury[J]Neurotrauma, 2020 Jan 15; 37(2): 389-396.
[22] Janotka M, Ostadal P. Biochemical markers for clinical monitoring of tissue perfusion[J]. Mol Cell Biochem, 2021, 476(3): 1313-1326. doi: 10.1007/s11010-020-04019-8
[23] Bitar ZI, Maadarani OS, El-Shably AM, et al. The Forgotten Hemodynamic(PCO2 Gap)in Severe Sepsis[J]. Crit Care Res Pract, 2020, 2020: 9281623.
[24] Mukai A, Suehiro K, Kimura A, et al. Comparison of the venousarterial CO2 to arterial-venous O2 content difference ratio with the venous-arterial CO2 gradient for the predictability of adverse outcomes after cardiac surgery[J]. Clin Monit Comput, 2020, 34(1): 41-53. doi: 10.1007/s10877-019-00286-z
[25] Roy TK, Secomb TW. Effects of impaired microvascular flow regulation on metabolism-perfusion matching and organ function[J]. Microcirculation, 2021, 28(3): e12673.
[26] Próspero AG, Soares GA, Moretto GM, et al. Dynamic cerebral perfusion parameters and magnetic nanoparticle accumulation assessed by AC biosusceptometry[J]. Biomed Tech(Berl), 2020, 65(3): 343-351. doi: 10.1515/bmt-2019-0089
-




 下载:
下载: