Latent Toxoplasma infection aggravates coagulation and renal dysfunction in elderly patients with sepsis by activating TLR-4
-
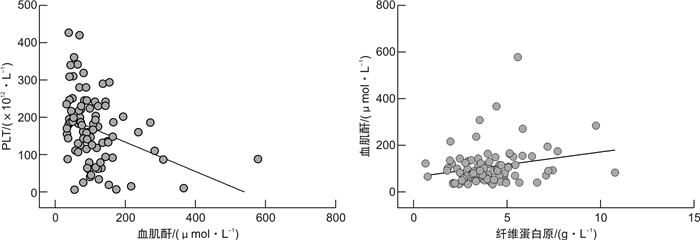
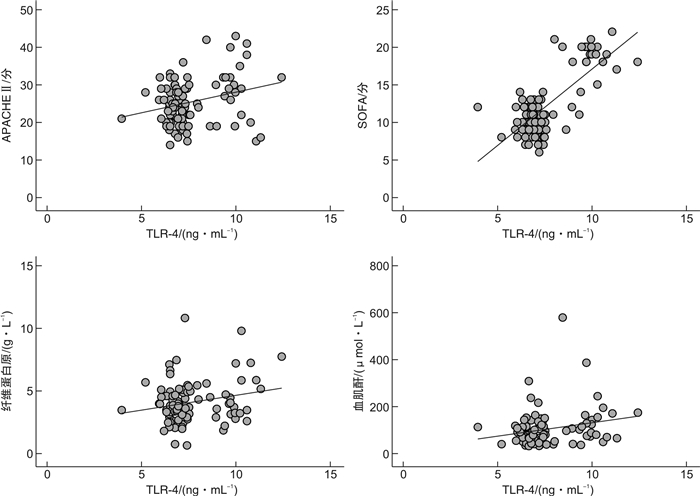
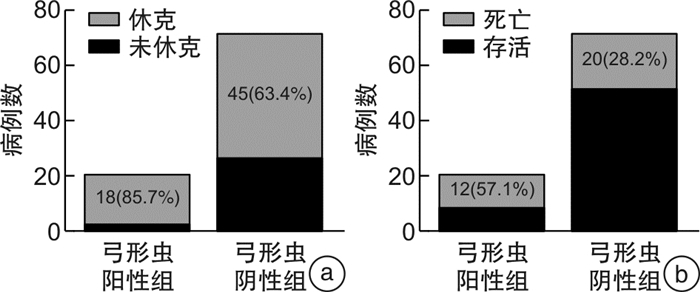
摘要: 目的 观察潜隐性弓形虫感染对老年脓毒症患者凝血功能和肾功能的影响,并探讨其可能的机制。方法 收集2015年1月—2019年12月入住无锡市中医医院重症医学科的92例老年脓毒症患者,根据弓形虫血清学IgG抗体是否阳性分为弓形虫阳性组(21例)和弓形虫阴性组(71例)。记录入院时APACHEⅡ评分、SOFA评分。观察两组患者血小板计数、凝血功能(PT、APTT、纤维蛋白原)、肾功能水平、休克的发生率及28 d病死率情况。检测两组患者血清炎症细胞因子(TLR-4、TNF-α、IL-6),分析炎症细胞因子与凝血功能、肾功能的相关性。结果 弓形虫阳性组入院时APACHEⅡ评分、SOFA评分高于弓形虫阴性组,差异有统计学意义(均P< 0.05); 弓形虫阳性组感染性休克发生率[85.7% (18/21)vs.63.4%(45/71)]及28 d病死率明显升高[57.1% (12/21)vs. 28.2%(20/71)]。弓形虫阳性组的脓毒症患者血小板计数较弓形虫阴性组低,但差异无统计学意义(P>0.05); 弓形虫阳性组PT、APTT水平与阴性组之间差异无统计学意义(均P>0.05); 弓形虫阳性组纤维蛋白原水平较阴性组明显升高,差异有统计学意义(P< 0.05); 弓形虫阳性组血肌酐水平较阴性组升高,差异有统计学意义(P< 0.05); 血肌酐水平与血小板计数呈负相关(r=-0.3447,P=0.0008)、与纤维蛋白原水平呈正相关(r=0.2309,P=0.0268)。弓形虫阳性组患者炎症细胞因子TLR-4、TNF-α、IL-6水平较弓形虫阴性组明显升高,差异有统计学意义(均P< 0.05)。进一步分析发现血清TLR-4水平与APACHEⅡ评分呈正相关(r=0.2721,P=0.0087)、与SOFA评分亦呈正相关(r=0.7589,P< 0.01); 血清TLR-4水平与纤维蛋白原水平呈正相关(r=0.2167,P=0.0038)、与血肌酐水平呈正相关(r=0.2280,P=0.0288)。结论 潜隐性弓形虫感染通过激活TLR-4,诱导炎症因子过度表达促进有害的全身性炎症反应及凝血功能紊乱,导致老年脓毒症患者肾脏损害及不良预后。Abstract: Objective To observe the effect of latent Toxoplasma infection on coagulation and renal function in elderly patients with sepsis and explore its possible mechanism.Methods The medical records of patients diagnosed with sepsis in ICU of Wuxi Hospital Affiliated to Nanjing University of Chinese Medicine from January 2015 to December 2019 were retrospectively analyzed. Patients were divided into positive group and negative group according to the serological antibody IgG of Toxoplasma gondii. Coagulation, platelet and renal function, incidence of shock and 28 days mortality were observed. The serum inflammatory cytokines(TLR-4, TNF-α, IL-6) and coagulation function(PT, APTT, fibrinogen) of the two groups were detected, and the correlation between inflammatory cytokines and coagulation function and renal function were analyzed.Results A total of 92 elderly patients with sepsis were collected, of which 21 were serologically positive and 71 were serologically negative. The APACHE Ⅱ score and SOFA score of positive group were higher than those of negative group(allP< 0.05). The incidence of septic shock and 28 days mortality were also significantly increased than the patients of negative group(18/21, 85.7% vs. 45/71, 63.4%; 12/21, 57.1% vs. 20/71, 28.2%). The platelet count of positive group had a downward trend than those of negative group, but there was no statistical difference(P>0.05). The PT and APTT levels of the positive group had no difference than the patients of negative group(allP>0.05); the fibrinogen level of the positive group was significantly higher than that of the negative group(P< 0.05); The serum creatinine level in patients of positive group was higher than the patients of negative group(P< 0.05). There was a negative correlation between serum creatinine level and platelet count(r=-0.3447,P=0.0008), and there was a positive correlation between serum creatinine level and fibrinogen(r=0.2309,P=0.0268). The serum inflammatory cytokines such as TLR-4, TNF-α and IL-6 were significantly higher than that of the negative group(allP< 0.05). Further analysis showed that, the serum level of TLR-4 was positively correlated with APACHEⅡ score and SOFA score(r=0.2721,P=0.0087;r=0.7589,P< 0.01); the serum level of TLR-4 was positively correlated with fibrinogen level(r=0.2167,P=0.0038).Conclusion Latent Toxoplasma gondii infection can induce excessive expression of inflammatory factors by activating TLR-4, promote harmful systemic inflammatory response and coagulation dysfunction, and lead to renal damage and poor prognosis in elderly patients with sepsis.
-
Key words:
- latent Toxoplasma infection /
- TLR-4 /
- sepsis /
- coagulation function /
- renal function
-

-
表 1 两组患者血小板计数及凝血功能
X±S 组别 例数 血小板/(×1012·L-1) PT/s APTT/s 纤维蛋白原/(g·L-1) 弓形虫阳性组 21 152.80±75.37 15.59±4.85 37.41±11.15 4.74±1.911) 弓形虫阴性组 71 175.00±94.34 15.89±9.56 37.90±12.36 3.85±1.58 与弓形虫阴性组比较,1)P < 0.05。 表 2 两组患者炎症介质
X±S 组别 例数 白细胞总数/(×109·L-1) CRP/(mg·L-1) TLR-4/(ng·mL-1) TNF-α/(ng·mL-1) IL-6/(ng·mL-1) 弓形虫阳性组 21 12.14±5.73 123.40±106.3 10.05±0.961) 531.20±61.491) 182.80±15.161) 弓形虫阴性组 71 12.42±6.89 101.00±85.82 6.92±0.79 419.70±79.53 100.00±12.98 与弓形虫阴性组比较,1)P < 0.05。 -
[1] Singer M, Deutschman CS, Seymour CW, et al. The third international consensus definitions for sepsis and septic shock(Sepsis-3)[J]. JAMA, 2016, 315(8): 801-810. doi: 10.1001/jama.2016.0287
[2] Tu H, Lai X, Li J, et al. Interleukin-26 is overexpressed in human sepsis and contributes to inflammation, organ injury, and mortality in murine sepsis[J]. Crit Care, 2019, 23(1): 290. doi: 10.1186/s13054-019-2574-7
[3] He F, Zhang C, Huang Q. Long noncoding RNA nuclear enriched abundant transcript 1/miRNA-124 axis correlates with increased disease risk, elevated inflammation, deteriorative disease condition, and predicts decreased survival of sepsis[J]. Medicine(Baltimore), 2019, 98(32): e16470.
[4] Popescu NI, Silasi R, Keshari RS, et al. Peptidoglycan induces disseminated intravascular coagulation in baboons through activation of both coagulation pathways[J]. Blood, 2018, 132(8): 849-860. doi: 10.1182/blood-2017-10-813618
[5] Busch D, Kapoor A, Rademann P, et al. Delayed activation of PPAR-β/δ improves long-term survival in mouse sepsis: effects on organ inflammation and coagulation[J]. Innate Immun, 2018, 24(4): 262-273. doi: 10.1177/1753425918771748
[6] 张志雄, 姜南, 赵忠岩, 等. 脓毒症所致急性肾损伤的发病机制研究进展[J]. 中国实验诊断学, 2019, 23(10): 1853-1857. doi: 10.3969/j.issn.1007-4287.2019.10.061
[7] Fox BA, Guevara RB, Rommereim LM, et al. Toxoplasma gondii Parasitophorous Vacuole Membrane-Associated Dense Granule Proteins Orchestrate Chronic Infection and GRA12 Underpins Resistance to Host Gamma Interferon[J]. mBio, 2019, 10(4): e00589-19. doi: 10.1128/mBio.00589-19
[8] Souza MC, Fonseca DM, Kanashiro A, et al. Chronic Toxoplasma gondii Infection Exacerbates Secondary Polymicrobial Sepsis[J]. Front Cell Infect Microbiol, 2017, 7: 116.
[9] 中华医学会重症医学分会. 中国严重脓毒症/脓毒性休克治疗指南(2014)[J]. 中华内科杂志, 2015, 54(6): 557-581.
[10] 刁一芮, 丁奇, 史渊源. 模式识别受体PRRs在急性肺损伤/急性呼吸窘迫综合征发病机制中的作用研究进展[J]. 中国免疫学杂志, 2022, 38(2): 227-233.
[11] Jekarl DW, Kim KS, Lee S, et al. Cytokine and molecular networks in sepsis cases: a network biology approach[J]. Eur Cytokine Netw, 2018, 29(3): 103-111. doi: 10.1684/ecn.2018.0414
[12] Alhusseiny SM, Saleh NE, El-Zayady WM, et al. Association between Toxoplasma gondii infection and coronary atherosclerosis[J]. Trans R Soc Trop Med Hyg, 2021, 115(10): 1190-1197. doi: 10.1093/trstmh/trab018
[13] Wang AW, Avramopoulos D, Lori A, et al. Genome-wide association study in two populations to determine genetic variants associated with Toxoplasma gondii infection and relationship to schizophrenia risk[J]. Prog Neuropsychopharmacol Biol Psychiatry, 2019, 92: 133-147. doi: 10.1016/j.pnpbp.2018.12.019
[14] Bhadra R, Gigley JP, Khan IA. The CD8 T-cell road to immunotherapy of toxoplasmosis[J]. Immunotherapy, 2011, 3(6): 789-801. doi: 10.2217/imt.11.68
[15] 郭晓芳, 梁培, 尤勇, 等. 腹腔感染相关脓毒症患者凝血功能障碍发病及危险因素分析[J]临床急诊杂志, 2022, 23(4): 260-264.
[16] 周醒, 谢剑锋, 杨毅. 脓毒症相关凝血功能障碍的机制及药物治疗进展[J]. 中华内科杂志, 2022, 61(2): 224-228.
[17] 李晋, 王燕, 曾亚薇, 等. 脓毒症相关凝血功能紊乱与免疫反应相关机制的研究进展[J]. 中华危重病急救医学, 2021, 33(12): 1519-1523.
[18] Anderberg SB, Luther T, Frithiof R. Physiological aspects of Toll-like receptor 4 activation in sepsis-induced acute kidney injury[J]. Acta Physiol(Oxf), 2017, 219(3): 573-588.
[19] Andonengi G, Kerfoot SM, McNagny K, et al. Platelets express functional Toll-like receptor-4[J]. Blood, 2005, 106(7): 2417-2423. doi: 10.1182/blood-2005-03-0916
[20] 蒋伟, 郑瑞强. 金属蛋白酶组织抑制剂-2联合胰岛素样生长因子结合蛋白7对脓毒症相关性急性肾损伤的临床应用价值研究进展[J]. 中华危重病急救医学, 2022, 34(1): 105-109.
[21] Jin YH, Li ZT, Chen H, et al. Effect of dexmedetomidine on kidney injury in sepsis rats through TLR4/MyD88/NF-κB/iNOS signaling pathway[J]. Eur Rev Med Pharmacol Sci, 2019, 23(11): 5020-5025.
[22] Yu Y, Li M, Su N, et al. Honokiol protects against renal ischemia/reperfusion injury via the suppression of oxidative stress, iNOS, inflammation and STAT3 in rats[J]. Mol Med Rep, 2016, 13(2): 1353-1360.
[23] Klenzak J, Himmelfarb J. Sepsis and the kidney[J]. Crit Care Clin, 2005, 21(2): 211-222.
-




 下载:
下载: