-
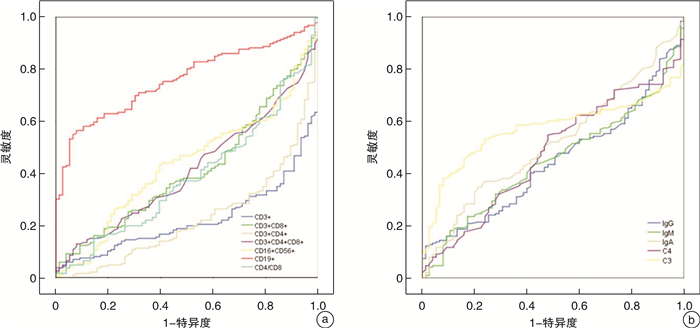
摘要: 目的 研究儿童脓毒性休克的免疫紊乱特点,评估外周血淋巴细胞亚群及体液免疫的改变能否为脓毒症患儿的病情预警及临床诊治提供参考。方法 选取昆明市儿童医院PICU 2018年1月—2020年12月收治的65例脓毒性休克患儿为脓毒性休克组,将同期121例脓毒症患儿为脓毒症组,76例健康体检儿童为对照组。收集研究对象的一般资料、淋巴细胞亚群、体液免疫、炎性指标、血生化等临床资料。采用统计软件SPSS 24.0对数据进行分析。结果 ① 对比分析3组的淋巴细胞亚群,发现T细胞、CD4+T细胞百分率在脓毒症组及对照组间随病情严重程度呈下降趋势,CD19+B细胞百分率随病情严重程度呈上升趋势,脓毒性休克组的CD16+CD56+百分率明显低于对照组,均差异有统计学意义(P<0.05)。②对比分析3组的体液免疫,发现脓毒性休克组的IgG、C3、C4明显低于对照组,均差异有统计学意义(P<0.05)。③通过ROC曲线评价免疫指标对脓毒症的诊断价值,结果显示CD19+B细胞百分率的曲线下面积为0.759,具有一定预测性。④通过logistic回归分析发现IgG和C3是影响脓毒症病情加重的危险因素,IgG和C3的浓度与脓毒症进展为脓毒性休克呈负相关(IgG:OR=0.827,P=0.003;C3:OR=0.133,P=0.002)。⑤通过Pearson相关性分析发现APACHEⅡ评分与CD3+CD4+T细胞百分率、CD4+/CD8+呈负相关,与CD3+CD8+T细胞百分率呈正相关;SOFA评分与CD4+/CD8+呈负相关,与CD3+CD8+T细胞百分率呈正相关;APACHEⅡ评分、SOFA评分均与C3、C4呈负相关。⑥脓毒性休克患儿的肝功能损伤与IgG、IgA呈正相关,与C3呈负相关;肾功能损伤与IgA呈正相关,与C3呈负相关;心肌损伤与C3、C4呈负相关;凝血功能障碍与C3、C4呈负相关;脓毒性休克患儿的器官功能障碍与淋巴细胞亚群不存在相关性。结论 ①儿童脓毒性休克及脓毒症均存在细胞免疫紊乱,且脓毒性休克的免疫紊乱更为严重;②随着脓毒症病情的加重,细胞免疫紊乱表现为CD4+T细胞、NK细胞逐渐衰竭、CD19+B细胞大量增殖活化;③脓毒性休克时存在B淋巴细胞功能障碍及补体系统紊乱,表现为IgG、C3、C4的浓度明显降低,且降低程度与SOFA评分、APACHEⅡ评分呈负相关;此外,IgG和C3降低是脓毒性休克发生的危险因素;④补体的过度激活和消耗与器官功能损伤密切相关; ⑤SOFA评分、APACHE Ⅱ评分对脓毒症、脓毒性休克患儿的病情评估和预后预测具有较高的价值。Abstract: Objective To study the characteristics of immune disorders in pediatric septic shock, and to evaluate whether the changes of peripheral blood lymphocyte subsets and humoral immunity can provide predictive ability for early recognition, clinical diagnosis and treatment of pediatric sepsis.Methods A total of 65 children with septic shock admitted to the PICU of Kunming Children's Hospital from January 2018 to December 2020 were selected as the experimental group, 121 children with sepsis and 76 healthy children during the same period were selected as the control group. The general information, lymphocyte subsets, humoral immunity, inflammatory indexes, blood biochemistry and other clinical data of the research subjects were collected. Statistical software SPSS 24.0 was used to analyze the data.Results ① The lymphocyte subsets of the experimental group and the control group were compared and analyzed. Results showed that the percentage of T cells and CD4+T cells decreased according with the severity of the disease between the sepsis and healthy children. The percentage of CD19+ increased according with the severity of the disease.The percentage of CD16+CD56+ in the septic shock group was significantly lower than that in the control group and the difference was statistically significant (P < 0.05). ②Humoral immunity of the experimental group and the control group was compared and results showed that the IgG, C3 and C4 of the septic shock group were significantly lower than those of the control group and the difference was statistically significant (P < 0.05). ③The diagnostic value of immune indexes for sepsis was evaluated by ROC curve. The results showed that the area under the curve of CD19+percentage was 0.759.④The results of logistic regression analysis, showed that IgG and C3 were the risk factors of the aggravation of sepsis, and the concentrations of IgG and C3 were negatively correlated with the progression of sepsis to septic shock(IgG: OR=0.827, P=0.003; C3: OR=0.133, P=0.002).⑤The results of Pearson correlation analysis, showed that APACHEⅡscore was negatively correlated with CD3+CD4+percentage, CD4+/CD8+, and positively correlated with CD3+CD8+percentage; SOFA score was negatively correlated with CD4+/CD8+, and positively correlated with CD3+CD8+percentage; APACHEⅡscore and SOFA score were negatively correlated with C3 and C4.⑥Liver function damage in children with septic shock was positively correlated with IgG, IgA, and negatively correlated with C3; renal function damage was positively correlated with IgA, and negatively correlated with C3; myocardial injury was negatively correlated with C3, C4;coagulation Dysfunction was negatively correlated with C3 and C4; there was no correlation between organ dysfunction and lymphocyte subsets inpediatric septic shock.Conclusion ① There are cellular immune disorders in pediatric septic shock and sepsis and the immune disorders in septic shock are more serious.②With the aggravation of sepsis, the cellular immune disorder is manifested as the exhaustion of CD4+T cells and NK cells and the proliferation of CD19+B cells.③There are obvious B lymphocyte dysfunction and complement system disorders in septic shock, which are manifested as the concentrations of IgG, C3, and C4 significantly reduced and the degree of reduction is negatively correlated with SOFA score and APACHEⅡscore; in addition, the reduction of IgG and C3 is a risk factor of the septic shock.④The excessive activation and consumption of complement is closely related to the damage of organ function.⑤SOFA score and APACHE Ⅱ score have obvious predictive ability in the disease severity assessment and prognosis prediction of patients with sepsis and septic shock.
-
Key words:
- septic shock /
- sepsis /
- immune disorder /
- lymphocyte subsets /
- humoral immunity
-

-
表 1 3组间淋巴细胞亚群及体液免疫的差异性分析
X±S 指标 脓毒性休克组(n=65) 脓毒症组(n=121) 对照组(n=76) F P CD3+/% 50.07±15.88 54.09±12.76 64.59±7.23 27.228 <0.001 CD3+CD8+/% 19.46±9.96 19.66±9.23 20.87±6.75 0.582 0.559 CD3+CD4+/% 26.64±10.96 30.01±9.29 38.91±8.58 32.788 <0.001 CD3+CD4+CD8+/% 0.28±0.35 0.39±0.86 0.33±0.25 0.662 0.517 CD16+CD56+/% 9.16±5.58 13.12±7.35 13.00±6.81 8.134 <0.001 CD19+/% 38.75±15.37 30.71±13.87 21.33±6.81 33.390 <0.001 CD4+/CD8+ 1.75±1.19 1.85±1.03 2.13±1.02 2.570 0.078 IgG /(g·L-1) 5.30±3.23 7.99±6.13 6.62±2.69 7.178 0.001 IgM /(g·L-1) 0.84±0.47 0.96±0.60 0.97±0.50 1.411 0.246 IgA /(g·L-1) 0.56±0.70 0.56±0.62 0.47±0.57 0.621 0.538 C4 /(g·L-1) 0.15±0.09 0.23±0.12 0.21±0.09 11.313 <0.001 C3 /(g·L-1) 0.71±0.40 1.14±0.44 0.91±0.22 27.120 <0.001 表 2 生存组和死亡组淋巴细胞亚群及体液免疫的差异性分析
X±S,M(Q) 指标 生存组(n=43) 死亡组(n=22) t/z P CD3+/% 48.90±14.24 52.34±18.83 -0.824 0.413 CD3+CD8+/% 17.00(9.72) 18.58(12.89) -1.422 0.149 CD3+CD4+/% 27.86±10.48 24.24±11.73 1.266 0.210 CD3+CD4+CD8+/% 0.20(0.24) 0.23(0.33) -0.167 0.537 CD16+CD56+/% 8.29(6.40) 7.57(6.86) -0.021 0.983 CD19+/% 40.31±13.09 35.70±19.05 1.020 0.316 CD4+/CD8+ 1.51(1.39) 1.20(0.82) -1.795 0.073 IgG /(g·L-1) 4.50(2.70) 4.40(2.83) -0.458 0.647 IgM /(g·L-1) 0.27(0.88) 0.29(0.57) 0.714 0.478 IgA /(g·L-1) 0.56±0.70 0.56±0.62 -0.583 0.560 C4 /(g·L-1) 0.16±0.09 0.13±0.09 1.322 0.191 C3 /(g·L-1) 0.77±0.44 0.61±0.31 1.764 0.083 表 3 免疫指标与临床指标的相关系数比较
指标 CD3+ CD3+CD8+ CD3+CD4+ CD3+CD4+CD8+ CD16+CD56+ CD19+ APACHEⅡ评分 -0.055 0.302 -0.403 0.038 0.181 -0.046 SOFA评分 0.132 0.337 -0.218 0.119 -0.077 -0.136 血小板减少 0.106 0.207 -0.178 0.275 -0.075 -0.095 肝功能损伤 0.197 0.243 -0.100 -0.035 -0.073 -0.214 肾功能损伤 -0.018 0.029 0.016 0.119 0 -0.007 凝血功能障碍 -0.026 -0.051 0.022 -0.094 -0.072 0.057 心肌损伤 0.079 0.119 -0.070 0.099 0.040 -0.060 胃肠功能紊乱 -0.218 -0.065 -0.194 0.085 0.134 0.169 指标 CD4+/CD8+ IgG IgM IgA C4 C3 APACHEⅡ评分 -0.462 -0.057 0.135 0.023 -0.250 -0.304 SOFA评分 -0.349 0.033 -0.004 0.066 -0.296 -0.421 血小板减少 -0.155 -0.006 -0.069 0.026 -0.139 -0.347 肝功能损伤 -0.206 0.271 0.193 0.282 -0.067 -0.287 肾功能损伤 -0.048 0.183 0.032 0.356 -0.183 -0.439 凝血功能障碍 -0.006 -0.179 -0.084 0.071 -0.322 -0.400 心肌损伤 -0.133 0.058 -0.123 0.067 -0.339 -0.407 胃肠功能紊乱 -0.111 -0.006 0.034 0.002 0.002 -0.086 -
[1] Carolin F, David MG, Peter S, et al. The global burden of paediatric and neonatal sepsis: a systematic review[J]. Lancet Resp Med, 2018, 6(3): 223-230. doi: 10.1016/S2213-2600(18)30063-8
[2] Singer M, Deutschman CS, Seymour CW, et al. The Third International Consensus Definitions for Sepsis and Septic Shock(Sepsis-3)[J]. JAMA, 2016, 315(8): 801-810. doi: 10.1001/jama.2016.0287
[3] 中国研究型医院学会休克与脓毒症专业委员会, 中国人民解放军重症医学专业委员会, 重症免疫研究协作组, 等. 脓毒症免疫抑制诊治专家共识[J]. 中华危重病急救医学, 2020, 32(11): 1281-1289. doi: 10.3760/cma.j.cn121430-20201123-00719
[4] Tian TW, Yong MY. Immunologic dissonance and its clinical significance in sepsis[J]. Med J Chinese PLA, 2017, 42(2): 95-102.
[5] 中华医学会儿科学分会急救学组, 中华医学会急诊医学分会儿科学组, 中国医师协会儿童重症医师分会. 儿童脓毒性休克(感染性休克)诊治专家共识(2015版)[J]. 中华儿科杂志, 2015, 53(8): 576-580. doi: 10.3760/cma.j.issn.0578-1310.2015.08.007
[6] 胡梓菡, 谢剑锋, 杨毅. 自然杀伤细胞在脓毒症免疫功能障碍中的研究进展[J]. 中华重症医学电子杂志(网络版), 2019, 5(2): 194-198. doi: 10.3877/cma.j.issn.2096-1537.2019.02.020
[7] Hotchkiss RS, Monneret G, Payen D. Sepsis-induced immunosuppression: from cellular dysfunctions to immunotherapy[J]. Nat Rev Immunol, 2013, 13(12): 862-874. doi: 10.1038/nri3552
[8] Holub M, Kluckova Z, Beneda B, et al. Changes in lymphocyte subpopulations and CD3+/DR+ expression in sepsis[J]. Clin Microbiol Infec, 2000, 6(12): 657-660. doi: 10.1046/j.1469-0691.2000.00175.x
[9] Jorge M, Raul DP, Eduardo R, et al. Clinical relevance of the severe abnormalities of the T cell compartment in septic shock patients[J]. Crit Care, 2009, 13(1): R26. doi: 10.1186/cc7731
[10] Ye Q, Shao W, Wang Q, et al. An imbalance of T cell subgroups exists in children with sepsis[J]. Microbes Infect, 2019, 21(8-9): 386-392. doi: 10.1016/j.micinf.2019.04.002
[11] Frattari A, Polilli E, Primiterra V, et al. Analysis of peripheral blood lymphocyte subsets in critical patients at ICU admission: A preliminary investigation of their role in the prediction of sepsis during ICU stay[J]. Int J Immunopath Ph, 2018, 32: 2058738418792310.
[12] 龙盼, 李响, 姜丽静, 等. 脓毒症休克患者临床免疫功能分析[J]. 重庆医学, 2017, 46(2): 198-200. doi: 10.3969/j.issn.1671-8348.2017.02.016
[13] Hotchkiss RS, Tinsley KW, Swanson PE, et al. Sepsis-induced apoptosis causes progressive profound depletion of B and CD4+T lymphocytes in humans[J]. J Immunol, 2001, 166(11): 6952-6963. doi: 10.4049/jimmunol.166.11.6952
[14] 曹雪涛. 医学免疫学[M]. 北京: 人民卫生出版社, 2013: 57-58.
[15] Manu S, Nicholas C, Benjamin P, et al. Endogenous IgG hypogammaglobulinaemia in critically ill adults with sepsis: systematic review and meta-analysis[J]. Intens Care Med, 2015, 41(8): 1393-1401. doi: 10.1007/s00134-015-3845-7
[16] Lee WL, Slutsky AS. Sepsis and Endothelial Permeability[J]. New Engl J Med, 2010, 363(7): 689-691. doi: 10.1056/NEJMcibr1007320
[17] Michaelsen TE, Sandlie I, Bratlie DB, et al. Structural difference in the complement activation site of human IgG1 and IgG3[J]. Scand J Immunol, 2009, 70(6): 553-564. doi: 10.1111/j.1365-3083.2009.02338.x
[18] Nordenfelt P, Waldemarson S, Linder A, et al. Antibody orientation at bacterial surfaces is related to invasive infection[J]. J Exp Med, 2012, 209(13): 2367-2381. doi: 10.1084/jem.20120325
[19] Shankar-Hari M, Spencer J, Sewell WA, et al. Bench-to-bedside review: Immunoglobulin therapy for sepsis-biological plausibility from a critical care perspective[J]. Crit Care, 2012, 16(2): 206.
[20] Bermejo-Martin JF, Rodriguez-Fernandez A, Herran-Monge R, et al. Immunoglobulins IgG1, IgM and IgA: a synergistic team influencing survival in sepsis[J]. J Intern Med, 2014, 276(4): 404-412. doi: 10.1111/joim.12265
[21] Raquel A, Eduardo T, Maria H, et al. Transcriptomic evidence of impaired immunoglobulin G production in fatal septic shock[J]. J Crit Care, 2014, 29(2): 307-309. doi: 10.1016/j.jcrc.2013.11.020
[22] 孟祥忠, 张晓宁, 朱宇. 重症脓毒症休克患者免疫状况的临床分析[J]. 实用临床医药杂志, 2018, 22(1): 4-6. https://www.cnki.com.cn/Article/CJFDTOTAL-XYZL201801002.htm
[23] 孙杭, 张宪伟, 潘伟. 补体消耗与脓毒症患儿病情严重程度的相关性[J]. 中华实用儿科临床杂志, 2017, 32(6): 425-429. doi: 10.3760/cma.j.issn.2095-428X.2017.06.007
[24] Vosylius S, Sipylaite J, Ivaskevicius J. Sequential organ failure assessment score as the determinant of outcome for patients with severe sepsis[J]. Croat Med J, 2004, 45(6): 715-720.
[25] 侯云静, 唐晟, 王彬, 等. 急性生理学及慢性健康评估Ⅱ系统在临床重症监护中的应用[J]. 中国医学装备, 2015, 12(1): 84-86. doi: 10.3969/J.ISSN.1672-8270.2015.01.027
[26] Maciej MM, John DL. The Role of Complement in Inflammatory Diseases From Behind the Scenes into the Spotlight[J]. Am J Pathol, 2007, 171(3): 715-727. doi: 10.2353/ajpath.2007.070166
[27] Maciej MM, Robert AD, John DL. Complexity of complement activation in sepsis[J]. J Cell Mol Med, 2008, 12(6a): 2245-2254. doi: 10.1111/j.1582-4934.2008.00504.x
[28] Amara U, Rittirsch D, Flierl M, et al. Interaction between the coagulation and complement system[J]. Adv Exp Med Biol, 2008, 632: 71-79.
[29] Maciej MM, Bo N, Kristina NE, et al. Complement and coagulation: strangers or partners in crime?[J]. Trends Immunol, 2007, 28(4): 184-192. doi: 10.1016/j.it.2007.02.006
[30] Levi M. Disseminated intravascular coagulation[J]. Crit Care Med, 2007, 35(9): 2191-2195. doi: 10.1097/01.CCM.0000281468.94108.4B
[31] Dhainaut J, Shorr AF, Macias WL, et al. Dynamic evolution of coagulopathy in the first day of severe sepsis: Relationship with mortality and organ failure[J]. Crit Care Med, 2005, 33(2): 341-348. doi: 10.1097/01.CCM.0000153520.31562.48
-

| 引用本文: | 杨雅, 肖曙芳, 王艳俊, 等. 儿童脓毒性休克免疫紊乱的临床特点[J]. 临床急诊杂志, 2022, 23(7): 469-475. doi: 10.13201/j.issn.1009-5918.2022.07.003 |
| Citation: | YANG Ya, XIAO Shufang, WANG Yanjun, et al. Clinical features of immune disorders in children with septic shock[J]. J Clin Emerg, 2022, 23(7): 469-475. doi: 10.13201/j.issn.1009-5918.2022.07.003 |
- Figure 1.



 下载:
下载: