-
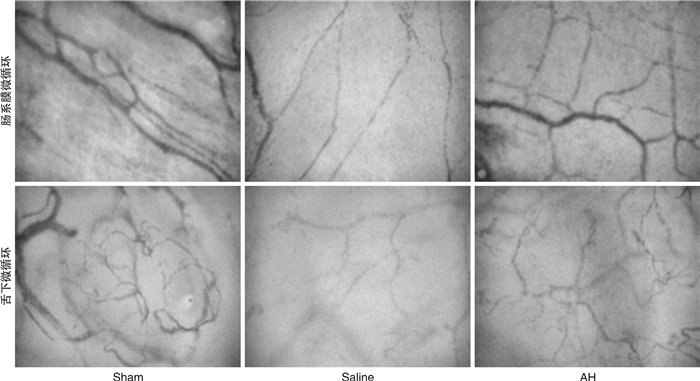
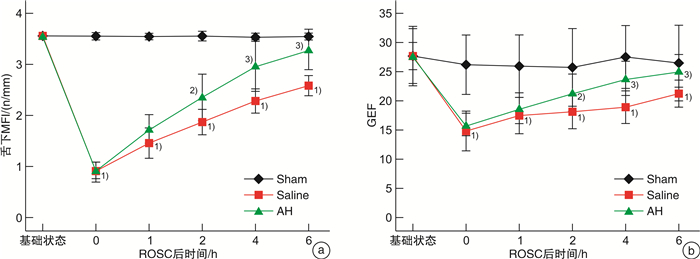
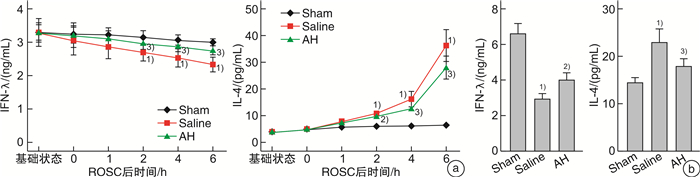
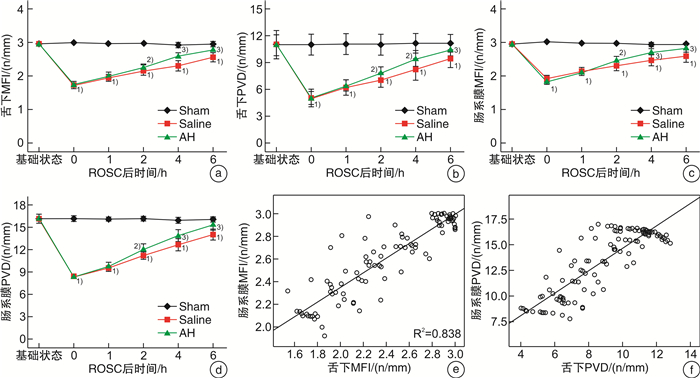
摘要: 目的 探讨复苏后机体微循环改变特点及氢溴酸山莨菪碱对机体的保护作用机制。方法 将24只北京白猪随机分为3组,即氢溴酸山莨菪碱组(AH组,8只),生理盐水组(Saline组,8只)及假手术组(Sham组,8只)。对AH组及Saline组动物应用电刺激诱导室颤,在室颤8 min后给予心肺复苏至恢复自主循环(recover the spontaneous circulation,ROSC)。Saline组复苏后仅给予盐水静点,AH组给予盐水+氢溴酸山莨菪碱静点。留取6个时间点(基础状态,ROSC即刻,ROSC1 h,ROSC2 h,ROSC4 h及ROSC 6 h)的动静脉血液,分别检测血气分析及细胞因子IL-4及IFN-γ,并检测各时间点的微循环指标PVD及MFI,同时测量连续心输出量(cardiac output,CO),全心射血分数(global ejection fraction,GEF)。分析3组之间上述指标的差异。结果 复苏后,PVD与MFI在肠道及舌下部位均明显下降(与基线和假手术组相比,P < 0.05)。自复苏后2 h开始,AH组与Saline组肠道PVD(12.09±0.75vs.11.23±0.53,14.53±0.69 vs.13.43±0.44,16.28±0.44 vs.15.53±0.50),肠道MFI(2.45±0.12 vs.2.31±0.16,2.70±0.10 vs.2.53±0.13,2.91±0.64 vs.2.67±0.14),舌下PVD(7.92±0.61 vs.7.09±0.84,9.52±0.92vs.8.29±1.25,11.10±0.97 vs.9.95±1.14),舌下MFI(2.28±0.06 vs.2.15±0.13,2.60±0.09 vs.2.30±0.35,2.85±0.03 vs.2.55±0.415)进行比较,均差异有统计学意义(P < 0.05)。肠道微循环血流量的变化与舌下微循环血流量的变化密切相关(PVD:r=0.864,P < 0.01;MFI:r=0.915,P < 0.01)。同时,复苏后机体心脏功能受损,细胞因子IFN-γ及IL-4水平异常,提示免疫失衡存在,AH组与Saline组相比,CO及GEF恢复更快且有利于维持IFN-γ/IL-4稳态。结论 肠道微循环在复苏后将出现功能损伤,其严重程度与心功能障碍成正相关,舌下微循环的测量可以反映肠道微循环的变化,复苏后细胞因子变化表现为免疫失衡,应用氢溴素山莨菪碱可以改善这种失衡,有利于心脏功能恢复,推测与该药物可以改善微循环有关。Abstract: Objective To investigate the characteristics of microcirculatory changes after resuscitation and the protective mechanism of anisodamine hydrobromide on the body.Methods Twenty-four Beijing white pigs were randomly divided into three groups: anisodamine hydrobromide group(AH group, 8 pigs), saline group(Saline group, 8 pigs) and sham operation group(Sham group, 8 pigs). Electric stimulation was used to induce ventricular fibrillation in AH group and Saline group, and cardiopulmonary resuscitation was given to recover the spontaneous circulation(ROSC) after 8 minutes ventricular fibrillation. The Saline group was only given saline after ROSC, and the AH group was given saline and anisodamine hydrobromide. The arterial and venous blood was collected at six time points: the baseline, ROSC instant, ROSC 1 h, ROSC 2 h, ROSC 4 h and ROSC 6 h, and the blood gas analysis and cytokine IL-4 and IFN-γ were measured respectively. The microcirculation indexes PVD and MFI, continuous cardiac output(CO) and global ejection fraction(GEF) were measured at each time point. Analyzed the differences of these indicators among the three groups.Results After resuscitation, PVD and MFI decreased significantly in both intestinal and sublingual regions(compared with baseline and sham operation groups, P < 0.05). From 2 hours after resuscitation, there were significant differences between AH group and Saline group: intestinal PVD(12.09±0.75 vs. 11.23±0.53, 14.53±0.69 vs. 13.43±0.44, 16.28±0.44 vs. 15.53±0.50, P < 0.05), intestinal MFI(2.45±0.12 vs. 2.31±0.16, 2.70±0.10 vs. 2.53±0.13, 2.91±0.64 vs. 2.67±0.14, P < 0.05), Sublingual PVD(7.92±0.61 vs. 7.09±0.84, 9.52±0.92 vs. 8.29±1.25, 11.10±0.97 vs. 9.95±1.14, P < 0.05) and sublingual MFI(2.28±0.06 vs. 2.15±0.13, 2.60±0.09 vs. 2.30±0.35, 2.85±0.03 vs. 2.55±0.415, P < 0.05). The change of intestinal microcirculation was closely related to the change of sublingual microcirculation(PVD: r=0.864, P < 0.01; MFI: r=0.915, P < 0.01). Meanwhile, after resuscitation, the cardiac function was damaged, and the change of levels of IFN-γ and IL-4 indicated immune imbalance. Compared with Saline group, CO and GEF in AH group recovered faster and AH can help body to maintain IFN-γ/IL-4 steady state.Conclusion The severity of intestinal microcirculation dysfunction after resuscitation is closely related to the cardiac function, the sublingual microcirculation can reflect the changes of intestinal microcirculation, and the changes of cytokines after resuscitation show an immune imbalance. Anisodamine hydrobromide can improve this imbalance, and repair the cardiac function, it can be inferred that these effects are related to the ability of this drug to improve microcirculation.
-

-
表 1 3组动物基础数据比较
项目 Sham组(n=8) Saline组(n=8) AH组(n=8) P 体重/kg 23.75±0.96 24.13±1.96 24.0±1.41 0.930 心率/(次/min) 101.75±5.85 105.38±6.63 101.63±5.53 0.418 MAP/mmHg 99.50±11.94 103.70±20.38 99.45±14.93 0.898 CO/(L/min) 3.60±0.22 3.62±0.25 3.68±0.28 0.310 pH 7.44±0.06 7.38±0.09 7.34±0.09 0.196 乳酸/(mmol/L) 2.15±0.44 2.37±0.87 2.34±0.65 0.873 表 2 复苏效果比较
项目 Saline组
(n=8)AH组
(n=8)t P 复苏时间/min 5.60±3.13 9.88±5.59 -1.885 0.08 除颤次数 1.38±0.52 1.63±0.52 -0.966 0.35 表 3 血流动力学指标与微循环参数的相关性分析
指标 舌下微循环 肠系膜微循环 MFI PVD MFI PVD CO 0.927 0.859 0.943 0.933 GEF 0.592 0.631 0.576 0.594 表 4 ROSC 6 h后数据比较
数据 Sham组(n=8) Saline组(n=8) AH组(n=8) 心率/(次/min) 105.5±6.61 118.25±11.83 113.0±9.80 MAP/mmHg 95.25±4.28 102.38±10.22 97.58±14.91 CO/(L/min) 3.54±0.07 2.77±0.43 3.44±0.50 pH 7.38±0.03 7.30±0.23 7.41±0.12 乳酸/(mmol/L) 1.98±0.39 4.79±0.41 3.51±0.90 -
[1] 冯梦云, 尚桂莲. 心脏骤停后低温治疗时间窗的研究进展[J]. 临床急诊杂志, 2021, 22(3): 216-221. doi: 10.13201/j.issn.1009-5918.2021.03.014
[2] Neumar RW, Nolan JP, Adrie C, et al. Post-cardiac arrest syndrome: epidemiology, pathophysiology, treatment, and prognostication. A consensus statement from the International Liaison Committee on Resuscitation(American Heart Association, Australian and New Zealand Council on Resuscitation, European Resuscitation Council, Heart and Stroke Foundation of Canada, InterAmerican Heart Foundation, Resuscitation Council of Asia, and the Resuscitation Council of Southern Africa); the American Heart Association Emergency Cardiovascular Care Committee; the Council on Cardiovascular Surgery and Anesthesia; the Council on Cardiopulmonary, Perioperative, and Critical Care; the Council on Clinical Cardiology; and the Stroke Council[J]. Circulation, 2008, 118(23): 2452-2483. doi: 10.1161/CIRCULATIONAHA.108.190652
[3] Piton G, Capellier G. Biomarkers of gut barrier failure in the ICU[J]. Curr Opin Crit Care, 2016, 22(2): 152-160.
[4] Coopersmith CM, Stromberg PE, Dunne WM, et al. Inhibition of intestinal epithelial apoptosis and survival in a murine model of pneumonia-induced sepsis[J]. JAMA, 2002, 287(13): 1716-1721. doi: 10.1001/jama.287.13.1716
[5] Caraballo C, Jaimes F. Organ Dysfunction in Sepsis: An Ominous Trajectory From Infection To Death[J]. Yale J Biol Med, 2019, 92(4): 629-640.
[6] Cinel I, Kasapoglu US, Gul F, et al. The initial resuscitation of septic shock[J]. J Crit Care, 2020, 57: 108-117. doi: 10.1016/j.jcrc.2020.02.004
[7] Haheim B, Kondratiev T, Dietrichs ES, et al. Comparison Between Two Pharmacologic Strategies to Alleviate Rewarming Shock: Vasodilation vs. Inodilation[J]. Front Med(Lausanne), 2020, 7: 566388.
[8] Wan F, Du X, Liu H, et al. Protective effect of anisodamine hydrobromide on lipopolysaccharide-induced acute kidney injury[J]. Biosci Rep, 2020, 40(7): BSR20201812. doi: 10.1042/BSR20201812
[9] Du X, Liu H, Yue Y, et al. Anisodamine Hydrobromide Protects Glycocalyx and Against the Lipopolysaccharide-Induced Increases in Microvascular Endothelial Layer Permeability and Nitric Oxide Production[J]. Cardiovasc Eng Technol, 2021, 12(1): 91-100. doi: 10.1007/s13239-020-00486-8
[10] Sheng CY, Gao WY, Guo ZR, et al. Anisodamine restores bowel circulation in burn shock[J]. Burns, 1997, 23(2): 142-146. doi: 10.1016/S0305-4179(96)00086-1
[11] Wang S, Li C, Ji X, et al. Effect of continuous compressions and 30: 2 cardiopulmonary resuscitation on global ventilation/perfusion values during resuscitation in a porcine model[J]. Crit Care Med, 2010, 38(10): 2024-2030. doi: 10.1097/CCM.0b013e3181eed90a
[12] Valenzuela TD, Roe DJ, Cretin S, et al. Estimating effectiveness of cardiac arrest interventions: a logistic regression survival model[J]. Circulation, 1997, 96(10): 3308-3313. doi: 10.1161/01.CIR.96.10.3308
[13] Goedhart PT, Khalilzada M, Bezemer R, et al. Sidestream Dark Field(SDF)imaging: a novel stroboscopic LED ring-based imaging modality for clinical assessment of the microcirculation[J]. Opt Express, 2007, 15(23): 15101-15114. doi: 10.1364/OE.15.015101
[14] Hutchings S, Watts S, Kirkman E. The Cytocam video microscope. A new method for visualising the microcirculation using Incident Dark Field technology[J]. Clin Hemorheol Microcirc, 2016, 62(3): 261-271. doi: 10.3233/CH-152013
[15] Coppel J, Bountziouka V, Martin D, et al. A comparison of the quality of image acquisition between two different sidestream dark field video-microscopes[J]. J Clin Monit Comput, 2021, 35(3): 577-583. doi: 10.1007/s10877-020-00514-x
[16] Verdant CL, De Backer D, Bruhn A, et al. Evaluation of sublingual and gut mucosal microcirculation in sepsis: a quantitative analysis[J]. Crit Care Med, 2009, 37(11): 2875-2881. doi: 10.1097/CCM.0b013e3181b029c1
[17] Dubin A, Edul VS, Pozo MO, et al. Persistent villi hypoperfusion explains intramucosal acidosis in sheep endotoxemia[J]. Crit Care Med, 2008, 36(2): 535-542. doi: 10.1097/01.CCM.0000300083.74726.43
[18] Fries M, Weil MH, Sun S, et al. Increases in tissue PCO2during circulatory shock reflect selective decreases in capillary blood flow[J]. Crit Care Med, 2006, 34(2): 446-452. doi: 10.1097/01.CCM.0000196205.23674.23
[19] Pranskunas A, Pilvinis V, Dambrauskas Z, et al. Early course of microcirculatory perfusion in eye and digestive tract during hypodynamic sepsis[J]. Crit Care, 2012, 16(3): R83. doi: 10.1186/cc11341
[20] Petitjeans F, Geloen A, Pichot C, et al. Is the Sympathetic System Detrimental in the Setting of Septic Shock, with Antihypertensive Agents as a Counterintuitive Approach? A Clinical Proposition[J]. J Clin Med, 2021, 10(19): 4569. doi: 10.3390/jcm10194569
[21] Moledina DG, Mansour SG, Jia Y, et al. Association of T Cell-Derived Inflammatory Cytokines With Acute Kidney Injury and Mortality After Cardiac Surgery[J]. Kidney Int Rep, 2019, 4(12): 1689-1697. doi: 10.1016/j.ekir.2019.09.003
[22] Lee HM, Ghill BK, Park E, et al. Changes in the Ratio of T Helper 1 to T Helper 2 Signature Cytokines in Patients Undergoing Living Donor Liver Transplantation Surgery: A Prospective Controlled Study[J]. Transplant Proc, 2018, 50(10): 3621-3625. doi: 10.1016/j.transproceed.2018.08.055
[23] Afzali B, Lombardi G, Lechler RI, et al. The role of T helper 17(Th17) and regulatory T cells(Treg)in human organ transplantation and autoimmune disease[J]. Clin Exp Immunol, 2007, 148(1): 32-46. doi: 10.1111/j.1365-2249.2007.03356.x
[24] Bretscher PA. On the mechanism determining the TH1/TH2 phenotype of an immune response, and its pertinence to strategies for the prevention, and treatment, of certain infectious diseases[J]. Scand J Immunol, 2014, 79(6): 361-376. doi: 10.1111/sji.12175
[25] Paul WE, Zhu J. How are T(H)2-type immune responses initiated and amplified?[J]. Nat Rev Immunol, 2010, 10(4): 225-235. doi: 10.1038/nri2735
[26] Gu W, Zhang Q, Li CS. Effect of Splenic Regulatory T-cell Apoptosis on the Postresuscitation Immune Dysfunction in a Porcine Model[J]. Chin Med J(Engl), 2016, 129(13): 1577-1583.
[27] 顾伟, 李春盛, 殷文朋, 等. 参附注射液对心肺复苏后心肌功能障碍猪转录因子T-bet和GATA-3表达的影响[J]. 中华危重病急救医学, 2015, 27 (3): 190-196. doi: 10.3760/cma.j.issn.2095-4352.2015.03.007
[28] Peberdy MA, Andersen LW, Abbate A, et al. Inflammatory markers following resuscitation from out-of-hospital cardiac arrest-A prospective multicenter observational study[J]. Resuscitation, 2016, 103: 117-124. doi: 10.1016/j.resuscitation.2016.01.006
[29] Jou C, Shah R, Figueroa A, et al. The Role of Inflammatory Cytokines in Cardiac Arrest[J]. J Intensive Care Med, 2020, 35(3): 219-224. doi: 10.1177/0885066618817518
[30] Adrie C, Adib-Conquy M, Laurent I, et al. Successful cardiopulmonary resuscitation after cardiac arrest as a "sepsis-like" syndrome[J]. Circulation, 2002, 106(5): 562-568. doi: 10.1161/01.CIR.0000023891.80661.AD
[31] Katsoulis IE, Balanika A, Sakalidou M, et al. Extensive colonic necrosis following cardiac arrest and successful cardiopulmonary resuscitation: report of a case and literature review[J]. World J Emerg Surg, 2012, 7(1): 35. doi: 10.1186/1749-7922-7-35
[32] 王世伟, 郭建, 朱长清. 微循环检测技术及其在急诊危重医学中的应用进展[J]. 临床急诊杂志, 2019, 20(2): 168-172. https://www.cnki.com.cn/Article/CJFDTOTAL-ZZLC201902019.htm
-

| 引用本文: | 董桂娟, 杨军, 赵鑫, 等. 氢溴酸山莨菪碱可改善复苏后机体微循环[J]. 临床急诊杂志, 2023, 24(8): 424-431. doi: 10.13201/j.issn.1009-5918.2023.08.007 |
| Citation: | DONG Guijuan, YANG Jun, ZHAO Xin, et al. Anisodamine hydrobromide can improve microcirculation after resuscitation[J]. J Clin Emerg, 2023, 24(8): 424-431. doi: 10.13201/j.issn.1009-5918.2023.08.007 |
- Figure 1a~1d.
- Figure 2.
- Figure 3.
- Figure 4.



 下载:
下载: