Model of cardiac arrest in type 2 diabetes mellitus ratsestablished by modified transcutaneous electrical stimulation on epicardium
-
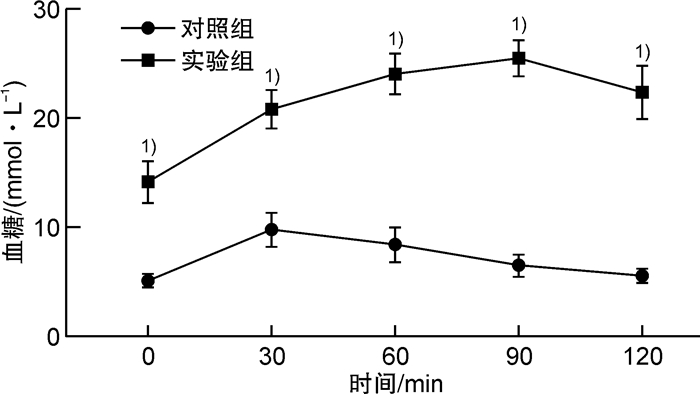
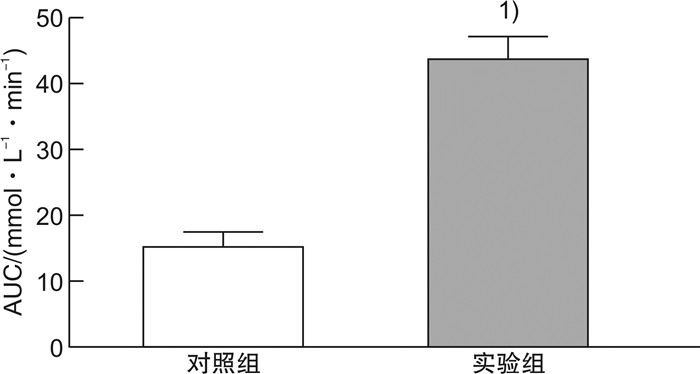
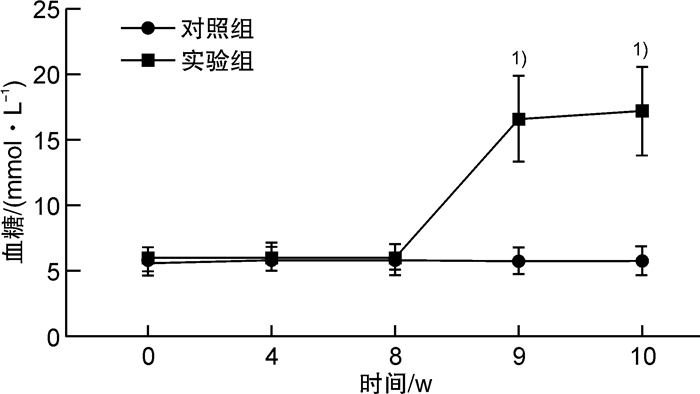
摘要: 目的探索构建2型糖尿病(T2DM)小动物心肺复苏模型的一种新方法,采用改良后的经心外膜电刺激诱导T2DM大鼠心室颤动,导致大鼠心搏骤停。方法选择健康的SPF级雄性Sprague-Dawley大鼠40只,体重为(250±10)g。其中10只大鼠作为对照组,给予普通饲料喂养并经腹腔注射同等剂量的柠檬酸钠溶液(PH=4.5);另30只大鼠给予高脂高糖饲料前期喂养,模拟糖尿病患者前驱期的肥胖、胰岛素抵抗和(或)葡萄糖不耐受状态,大鼠T2DM造模成功后,选用针灸针作为导电极,采用定量化定位点经胸壁刺入针灸针至达心外膜,持续交流电刺激诱导心室颤动并维持6 mins,然后给予心肺复苏术:呼吸机辅助通气、胸外心脏按压、静脉推注肾上腺素以及电除颤等。诱颤的电流强度为0.5~1.0 mA,电刺激总时间为3 min,电刺激后心电监护均显示心室颤动,心肺复苏后大鼠均恢复自主心率,实验结束后解剖尸体,肉眼观察未发现心外膜出血点和肺部淤血。结果24只大鼠造模2周后的血糖为(17.1±3.4)mmol/L,体重为(468.7±10.4)g,符合T2DM。有效电刺激开始后,本实验24只T2DM大鼠全部成功诱发心搏骤停。诱颤的电流强度为(0.6±0.1)mA,电刺激后诱发出现心搏骤停的时间为(20±6)s,室颤导致的心搏骤停的总时间为6 mins,心肺复苏时间为8 mins,除颤次数(3±2)次。实验结束后解剖尸体,肉眼观未发现心外膜出血点和肺部淤血。结论采用改良后经心外膜诱颤建立2型糖尿病大鼠心搏骤停/心肺复苏模型的操作简单可行,模型稳定,能够满足2型糖尿病患者心搏骤停/心肺复苏基础研究的需要。Abstract: ObjectiveTo explore a new method for constructing a small animal cardiopulmonary resuscitation model of type 2 diabetes mellitus(T2DM), using improved transepicardial electrical stimulation to induce ventricular fibrillation in T2DM rats, resulting in cardiac arrest in rats.MethodsThe experimental site was in the Cardiopulmonary Resuscitation Laboratory of the Second Affiliated Hospital of Anhui Medical University. A total of forty healthy male Sprague-Dawley rats, weighing(250±10)g, were selected. Ten of them were in the control group. The other 30 rats were fed with high-fat and high-sugar diet in the prodromal phase to simulate obesity, insulin resistance, and/or glucose intolerance of diabetic patients. The acupuncture needle was used as the lead electrode. Pierce the acupuncture needle through the chest wall to the epicardium at the quantitative positioning point. Ventricular fibrillation was induced by continuous alternating current stimulation and maintained for 6 minutes, and then cardiopulmonary resuscitation was performed, including ventilator-assisted ventilation, chest compressions, intravenous injection of epinephrine, and electrical defibrillation.The current intensity of the induced fibrillation was 0.5-1.0 mA, and the total time of electrical stimulation was 3 minutes. After electrical stimulation, the ECG monitoring of all rats showed ventricular fibrillation. After cardiopulmonary resuscitation, all rats recovered their voluntary heart rate. No macroscopic subepicardial hemorrhage and marked pulmonary congestion was found at the post-mortem autopsy.ResultsTwo weeks after STZ establishment, the blood glucose of 24 rats was (17.0±3.3)mmol/L, and the body weight of them was (468.7±10.4)g, meeting the standards of T2DM rats. After the effective electrical stimulation, cardiac arrest was successfully induced in all 24 T2DM rats. The current intensity of induced fibrillation is(0.6±0.1)mA, the time to induce cardiac arrest after electrical stimulation is(20±6)s, the total time of cardiac arrest induced by ventricular fibrillation is 6 min, the CPR time is 8 minutes, and the number of defibrillation is (3±2)times. No macroscopic subepicardial hemorrhage and obvious pulmonary congestion was found in the autopsy.ConclusionThe improved method of inducing cardiac arrest/cardiopulmonary resuscitation model in T2DM rats through transcutaneous electrical stimulation on epicardium is simple and feasible, and the model is stable, which can satisfy the needs of basic research on cardiac arrest/cardiopulmonary resuscitation in T2DM patients.
-
Key words:
- cardiac arrest /
- electrical epicardium stimulation /
- type 2 diabetes mellitus /
- rat
-

-
表 1 2组大鼠不同时间点体重变化情况
X±S 组别 第0周 第4周 第8周 第9周 第10周 对照组 250.5±4.7 324.3±7.11) 390.2±10.81) 405.3±9.61) 416.3±8.21) 实验组 249.8±5.62) 392.8±9.21)2) 512.7±8.11)2) 472.7±10.81)2) 468.7±10.42) 与同组上一测量时间点比较,1)P < 0.05;与对照组比较,2)P < 0.05。 表 2 大鼠诱发CA过程中各项指标变化
X±S 指标 数值 数量/只 24 体重/g 468.7±10.4 基线MAP/mmHg 419±25 基线HR/(次·min-1) 122±7 诱颤电流/mA 0.6±0.1 诱颤时间/s 20±6 按压深度/cm 1.1±0.1 除颤次数/次 3±2 -
[1] Kucharska-Newton AM, Couper DJ, Pankow JS, et al. Diabetes and the risk of sudden cardiac death, the Atherosclerosis Risk in Communities study[J]. Acta Diabetol, 2010, 47(Suppl 1): 161-168.
[2] Walker AM, Cubbon RM. Sudden cardiac death in patients with diabetes mellitus and chronic heart failure[J]. Diab Vasc Dis Res, 2015, 12(4): 228-233. doi: 10.1177/1479164115573225
[3] Jung YH, Lee BK, Jeung KW, et al. Association between Achievement of Estimated Average Glucose Level and 6-Month Neurologic Outcome in Comatose Cardiac Arrest Survivors: A Propensity Score-Matched Analysis[J]. J Clin Med, 2019, 8(9): 1480.
[4] Liu CH, Hua N, Fu X, et al. Metformin regulates atrial SK2 and SK3 expression through inhibiting the PKC/ERK signaling pathway in type 2 diabetic rats[J]. BMC Cardiovasc Disord, 2018, 18(1): 236. doi: 10.1186/s12872-018-0950-x
[5] Kim YG, Roh SY, Han KD, et al. Hypertension and diabetes including their earlier stage are associated with increased risk of sudden cardiac arrest[J]. Sci Rep, 2022, 12(1): 12307. doi: 10.1038/s41598-022-16543-2
[6] 杜兰芳, 李昭屏, 马青变. 亚低温对于心搏骤停患者复苏后心脏功能的影响[J]. 临床急诊杂志, 2019, 20(1): 33-35. https://www.cnki.com.cn/Article/CJFDTOTAL-ZZLC201901007.htm
[7] 刘国祥, 朱长清, 王世伟, 等. 心搏骤停后综合征相关急性胃肠损伤的研究进展[J]. 临床急诊杂志, 2021, 22(9): 634-640.
[8] Heydemann A. An Overview of Murine High Fat Diet as a Model for Type 2 Diabetes Mellitus[J]. J Diabetes Res, 2016, 2016: 2902351.
[9] Ocak U, Ocak PE, Huang L, et al. Inhibition of PAR-2 Attenuates Neuroinflammation and Improves Short-Term Neurocognitive Functions Via ERK1/2 Signaling Following Asphyxia-Induced Cardiac Arrest in Rats[J]. Shock, 2020, 54(4): 539-547. doi: 10.1097/SHK.0000000000001516
[10] López J, Fernández SN, González R, et al. Different Respiratory Rates during Resuscitation in a Pediatric Animal Model of Asphyxial Cardiac Arrest[J]. PLoS One, 2016, 11(9): e0162185. doi: 10.1371/journal.pone.0162185
[11] Sharp WW, Beiser DG, Fang YH, et al. Inhibition of the mitochondrial fission protein dynamin-related protein 1 improves survival in a murine cardiac arrest model[J]. Crit Care Med, 2015, 43(2): e38-47.
[12] Liu Y, Wang P, Wen C, et al. Endovascular hypothermia improves post-resuscitation myocardial dysfunction by increasing mitochondrial biogenesis in a pig model of cardiac arrest[J]. Cryobiology, 2019, 89(1): 6-13.
[13] Shah AR, Khan MS, Hirahara AM, et al. A real-time system for selectively sensing and pacing the His-bundle during sinus rhythm and ventricular fibrillation[J]. Biomed Eng Online, 2020, 19(1): 19.
[14] 左艳芳, 宋凤卿, 陈蒙华, 等. WIN55, 212-2在心肺复苏后对神经细胞凋亡的作用[J]. 中华急诊医学杂志, 2016, 25(4): 455-459. https://cdmd.cnki.com.cn/Article/CDMD-10422-1018106319.htm
[15] Chen MH, Liu TW, Xie L, et al. Ventricular fibrillation induced by transoesophageal cardiac pacing: a new model of cardiac arrest in rats[J]. Resuscitation, 2007, 74(3): 546-551.
[16] Budhram GR, Mader TJ, Lutfy L, et al. Left ventricular thrombus development during ventricular fibrillation and resolution during resuscitation in a swine model of sudden cardiac arrest[J]. Resuscitation, 2014, 85(5): 689-693.
[17] Lin JY, Liao XX, Li H, et al. Model of cardiac arrest in rats by transcutaneous electrical epicardium stimulation[J]. Resuscitation, 2010, 81(9): 1197-1204.
-




 下载:
下载: